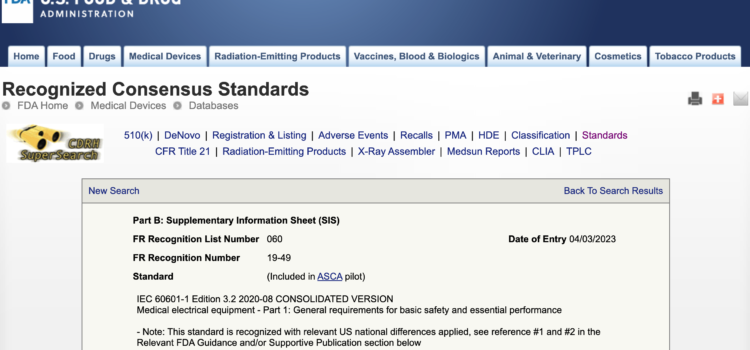
Great News!!!! Effective 19 September 2023 FDA’s Accreditation Scheme for Conformity Assessment (ASCA) Program converted from a pilot program after 3 years to a permanent program, the agency announced. The transition is authorized under the Medical Device User Fee Amendments
FDA’s ASCA Program Becomes Permanent