Draft EUDAMED roadmap (of modules*) was published on 20 Oct 2023 with updated timelines for each module and mandatory use dates for the MDR & IVDR.
Draft EUDAMED Roadmap of Modules


Draft EUDAMED roadmap (of modules*) was published on 20 Oct 2023 with updated timelines for each module and mandatory use dates for the MDR & IVDR.
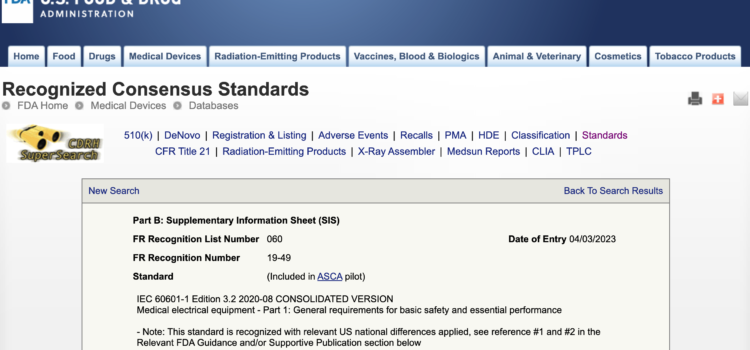
On April 3, 2023 FDA added IEC 60601-1 Edition 3.2 2020-08 CONSOLIDATED VERSION Medical electrical equipment – Part 1: General requirements for basic safety and essential performance to their list of Recognized Consensus Standards as Recognition # 19-49 in Recognition List

The FDA on May 30, 2022, same day as when the recent AAMI 60601 Amendments were Recognized, added the Interpretation Sheet 1 (ISH1) – Amendment 1 – on Essential Performance (Clause 4.3) and Single Fault Condition (Clause 4.7) based on

The FDA in a little over a month on 30 May 2022 (that is swift) has updated the Recognized Consensus database for the AAMI 60601 Amendmended Standards that are referenced in my previous post of May 13, 2022. The AAMI

After a delay of over a year since the original US Technical Advisory Group IEC/SC62A & specific technical committees — Electrical Safety, Electromagnetic Compatibility, Alarms, and Home Care and EMS Environments committees — vote and an additional vote for each of