FDA Updated Its Recognized Consensus Standards db

Key themes: IEC 60601/80601, IEC 61326-2-6, ISO 15223-1 A1:25, & ASCA considerations
In late December 2025, the FDA expanded its Recognized Consensus Standards Database, adding more than 100 new or updated standards. Some are eligible for use under the ASCA Program, while others carry important implications for design, testing, labeling, and overall regulatory strategy.
Regulatory expectations around standards are shifting earlier in the product lifecycle. Standards are no longer something teams can simply “handle at test.” Reviewers increasingly expect standards interpretation, risk decisions, and test strategies to be visible and justified during design reviews and technical documentation development. This reinforces why understanding not just which standards are recognized, but how they are recognized and applied, matters more than ever.
What the latest FDA update included
The scope of this database expansion spans a wide range of medical device technologies, including medical electrical equipment, IVDs, software-driven devices, imaging systems, interoperability standards, and labeling-related standards.
Beyond simply identifying newly recognized standards, the changes highlight how recognition status, ASCA eligibility, and extent of recognition can materially affect how conformity is demonstrated, how evidence is presented, and how submissions are reviewed.
Over the past year, I’ve been sharing what I’m seeing based on feedback gathered from many sources, including my Eisner Safety Consultants experts and discussions across the community with regulators, standards developers, test labs, and manufacturers. The goal is practical clarity: what changed, what it means, and what teams should do next as they navigate evolving standards expectations and regulatory requirements.
IEC 60601 / 80601 standards in the update
For manufacturers of medical electrical equipment, the December 22, 2025 update includes several standards from the IEC 60601 and 80601 series. These standards remain central to demonstrating basic safety and essential performance for medical electrical equipment. However, recognition alone does not mean every clause or requirement is fully accepted without qualification.
Standards called out in the December 22, 2025 update
(from the FDA database extract)
* Partial recognition. The following part(s) of the standard is (are) not recognized:
Subclause 202.8.101 “IMMUNITY test setup” – Reference to table height (>= 0,4 m) for IMMUNITY testing in Figure 202.101.
Refer to Rationale for Recognition for explanation of partial recognition.
EMC context within the IEC 60601 / 80601 framework
IEC 60601-1-2 remains the primary EMC standard within the IEC 60601 series. However, other IEC 60601 standards can and do include EMC-related requirements. Applicability must be assessed standard by standard, based on the specific requirements of each applicable document.
This context becomes especially important when considering how EMC expectations are addressed across different standard families, including IVD-specific EMC requirements discussed next.
IVD EMC and IEC 61326-2-6
A notable change reflected in the FDA Recognized Consensus Standards Database is the inclusion of IEC 61326-2-6 Edition 4 as a Recognized Consensus Standard, with ASCA eligibility under the ASCA Program, effective July 28, 2025.
This is significant because IEC 61326-2-6 is the primary EMC standard used to demonstrate electromagnetic compatibility for IVD medical equipment, and its inclusion under ASCA directly affects how EMC conformity evidence may be presented in FDA submissions.
However, ASCA eligibility alone does not guarantee streamlined review.
For manufacturers intending to rely on ASCA pathways for IVD EMC:
- Test lab scope must be verified as active for the specific standards and applicable editions being claimed.
- If a standard or edition is not within a laboratory’s ASCA-accredited scope,
manufacturers cannot rely on an ASCA Summary Test Report for that standard.
- In those cases, full EMC test reports remain necessary, and FDA review timelines are typically longer.
As of early January 2026, there are no ASCA-accredited laboratories that reflect IEC 61326-2-6 Edition 4 within their active scope. This creates a practical timing and planning consideration for manufacturers preparing IVD submissions that intend to leverage ASCA.
This gap does not change the applicability of IEC 61326-2-6 itself, but it does affect how conformity evidence can be packaged and reviewed until laboratory scopes are updated.
ISO 15223-1 Amendment 1:2025 and labeling implications
Authorized Representative symbol changes and labeling impact
ISO 15223-1 Amendment 1:2025 introduces a substantive correction to the requirements for the Authorized Representative symbol that has direct regulatory and labeling implications across multiple markets.
This change is implemented through a revision to Clause 5.1.2, which governs how the Authorized Representative symbol is applied.
Clause 5.1.2 – prior to Amendment 1:2025
“This symbol shall be accompanied by the name and address of the authorized representative, adjacent to the symbol.”
Under this wording:
- The symbol itself did not explicitly encode the jurisdiction
- The convention of using “EC REP” persisted in practice, even after the transition from the MDD & IVDD to EU MDR and IVDR
- This created ambiguity for non-EU markets and for products marketed globally under different regulatory frameworks
Clause 5.1.2 – after Amendment 1:2025
“The [XX] text of the symbol shall be replaced by either the two-letter country code or the three-letter country code defined in ISO 3166-1 or other text required by the authority having jurisdiction.
This symbol shall be accompanied by the name and address of the authorized representative adjacent to the symbol.”
Key changes introduced by this amendment:
- The Authorized Representative symbol must identify the jurisdiction represented
- The jurisdiction identifier is no longer implicit or assumed
- Country-specific representation is now explicitly required using ISO 3166-1 codes or authority-mandated text
This change aligns the symbol requirements with:
- EU MDR and IVDR expectations for EU Countries
- Non-EU regulatory frameworks that require local authorized representation
- Global labeling clarity for multi-market products
Practical application of the revised requirement
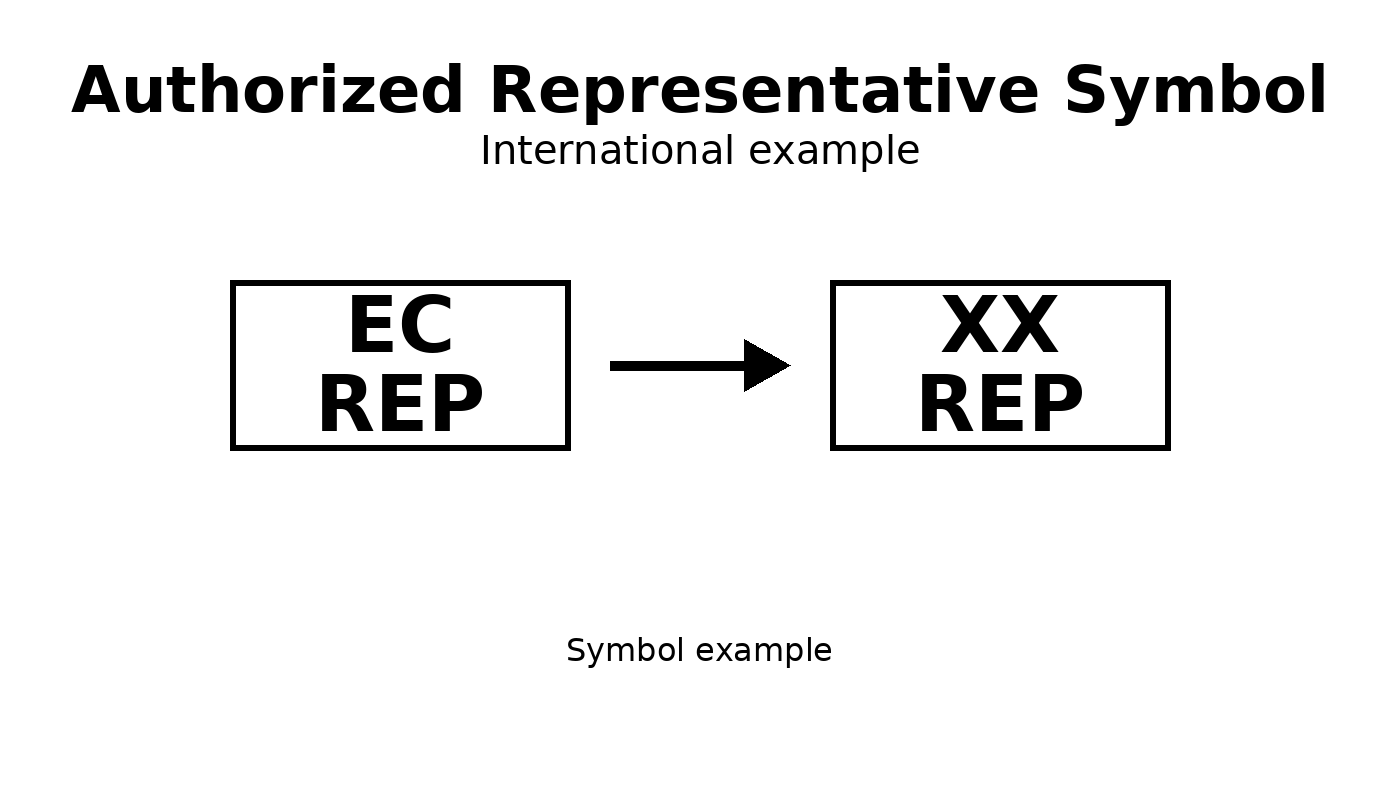
International example
Used for countries outside the EU, such as Switzerland or the United Kingdom.
Examples:
- CH REP for Switzerland
- UK REP for the United Kingdom
Convert from EC Rep to International two-letter country code Authorized Representative Symbol
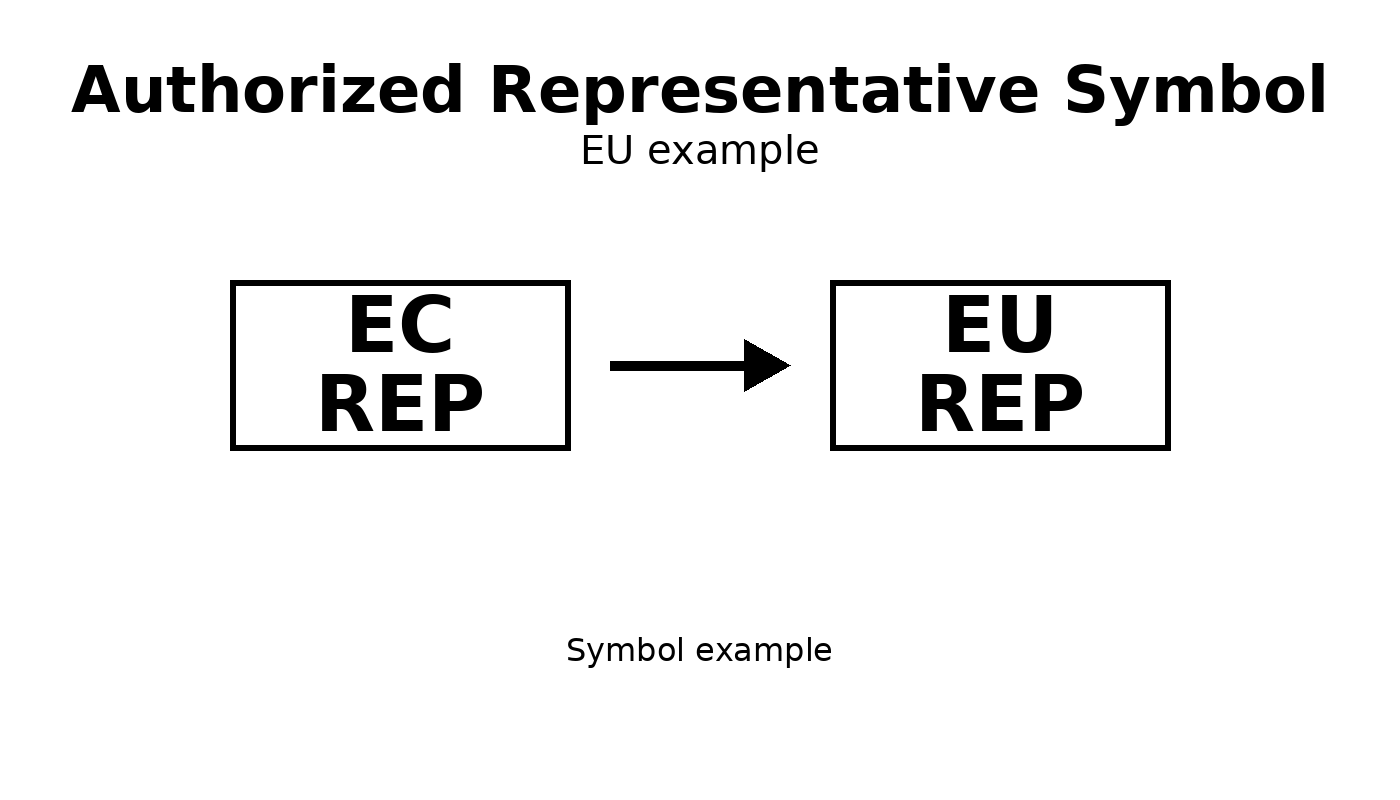
EU example
Used for EU Member States.
Convert from EC Rep to EU Rep Authorized Representative Symbol
Labeling and change-control implications
Symbol changes cascade into artwork, IFUs, packaging, and labeling change control. This affects and has significant impact on:
- Device labels
- IFUs
- Packaging artwork
- Labeling control procedures
These changes must be assessed and implemented through formal labeling change processes, not treated as isolated graphic updates.
Need help turning standards updates into practical actions?
My team & I at Eisner Safety Consultants supports medical device, diagnostic, and combination product manufacturers by translating standards into practical design inputs, test strategies, and documentation approaches aligned with FDA and other market expectations.
Keep the conversation going on LinkedIn
Add your perspective to the discussion on the December FDA standards update, including ASCA, IEC 60601/80601, IVD EMC,
ISO 15223-1 A1 & how these changes play out in practice.
Dec 22, 25 FDA db update (includes info on AAMI CR515:2025)
Recent FDA Recognized Consensus Standards Update
What Medical Device Manufacturers Should Know
Related technical discussion
High-engagement discussion on IVD EMC & ASCA implications:
IEC 61326-2-6 (IVD EMC) | 55+ comments
Reference materials
FDA Recognized Consensus Standards Database:
December 22, 2025 (all 100+ standards)
ASCA Eligible Standards List: December 22, 2025
