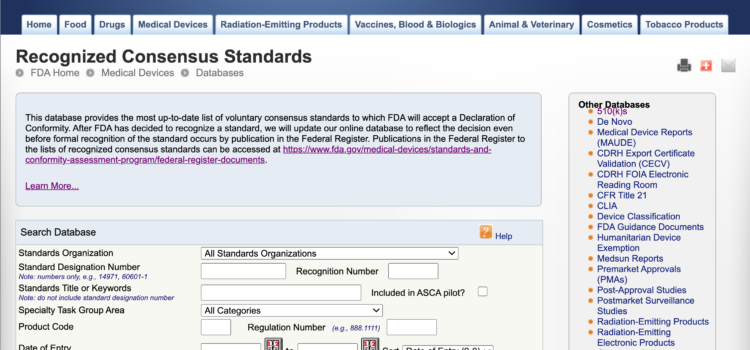
Updated 22 Dec 2020 – In the last couple of years, the FDA has started to post the standards updates on their Recognized Consensus Standards db prior to publishing in the Federal Register (FR). This year, being the year we
FDA Finally Adds to Fall 2020 Recognized Consensus Standards db for Cycle Update