The FDA has recently only Partially Recognized the IEC 61326-1:2020 & IEC 62316-2-6:2020 IVD EMC standards.
What does that mean to IVD EMC tests?
We have done a detailed analysis that has been confirmed by FD’s EMC experts to let you know what EMC tests you need to conduct in order to meet both FDA and EU requirements for IVD equipment.
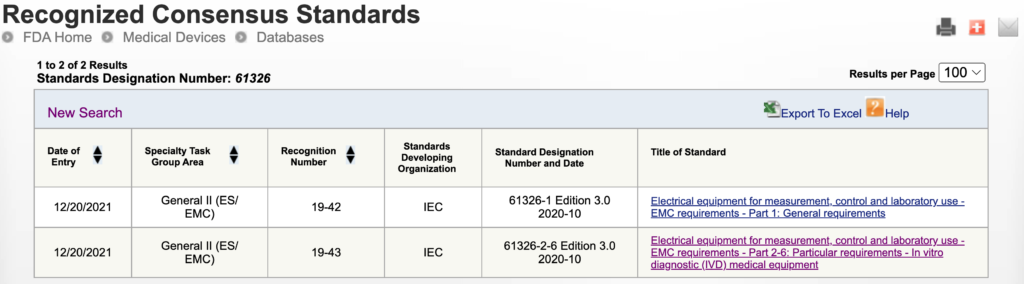
If you have IVD devices that you are submitting to the FDA you will need to be aware of these requirements. Rely on our experts for this analysis. We have had discussions with the FDA to understand the issues so we can explain this information.
You can obtain this analysis for only $1,500.00 by completing the form below.
